
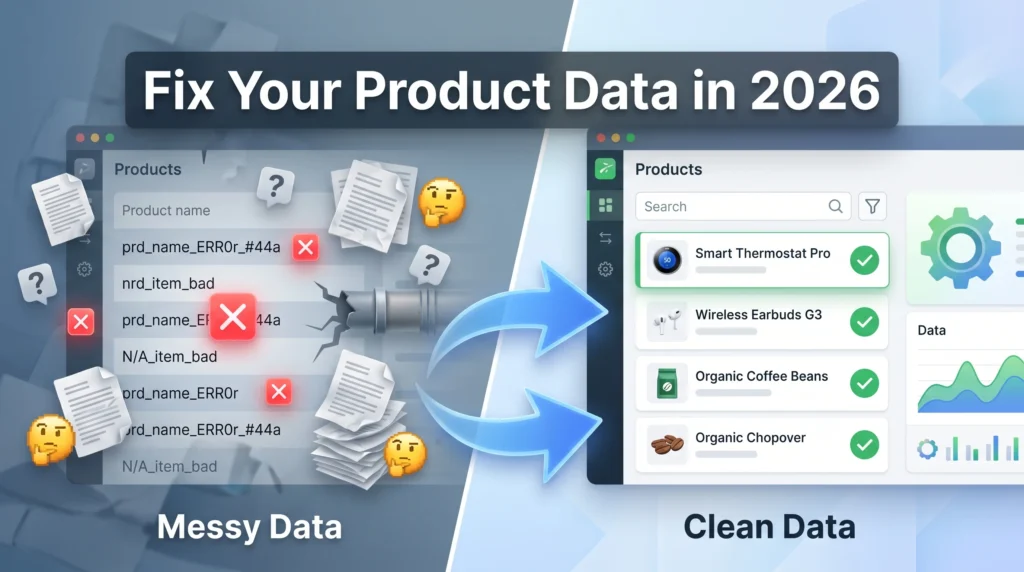
Your product catalog is costing you money right now, and you probably do not know it.
Duplicate entries, broken search results, failed ERP integrations, and compliance failures all trace back to one root cause: dirty product names. If you are responsible for a product database, an e-commerce catalog, or a healthcare inventory system, product name cleaning best practices are not optional in 2026. They are surviving.
This guide gives you a battle-tested, current framework updated for 2026’s AI-driven data landscape to clean, standardize, and future-proof your product naming data.
Let’s get into it.
Product name cleaning is the process of identifying, correcting, and standardizing product names across a dataset or catalog so that every entry is accurate, consistent, and usable.
Think of it as editing a messy spreadsheet, but at scale. You remove extra spaces, fix capitalization, expand or align abbreviations, and ensure that “Paracetamol 500mg Tab”, “paracetamol 500 mg tablet”, and “PARA 500MG TB” are all recognized as the same product.
This applies across industries:
In 2026, the stakes are higher. Gartner research projects that organizations will abandon 60% of AI initiatives due to insufficient data quality. Product name cleaning is no longer just a data hygiene task; it is an AI readiness requirement.
For a foundational understanding of how naming rules work at the brand and catalog level, explore these brand name normalization rules that apply directly to product catalog standardization.
The cost of dirty data has never been higher. McKinsey research shows that poor data quality leads to a 20% drop in productivity and a 30% rise in operating costs, for the average enterprise, which translates to roughly $12.9 million lost per year in data quality failures alone.
Here is what bad product names specifically cause:
| Problem | Business Impact |
|---|---|
| Duplicate product entries | Inflated inventory counts, inaccurate procurement |
| Inconsistent capitalization | Broken search filters, poor user experience |
| Unauthorized special characters | System errors, failed API integrations |
| Abbreviation mismatches | Wrong item picked, packed, or shipped |
| Non-standard formatting | Failed ERP syncs, regulatory submission errors |
| Semantic duplicates | AI models trained on wrong product classifications |
Clean product names deliver the opposite:
In healthcare, the impact goes beyond operational efficiency. A misspelled drug name or inconsistently labeled supply can trigger a serious clinical error. Clean data here is not just good practice; it is patient safety.
Follow this six-step process whether you are cleaning 500 product names or 5 million.
Pull a full export of your product catalog. Use a data profiling tool to surface:
@, #, *, &)In 2026, AI-powered profiling tools like Atlan and Ovaledge can automate most of this audit step, flagging problem records across millions of rows in minutes.
Before you touch a single record, write down your rules. Document:
Version this document. Call it “Product Naming Standard v1.0” from day one.
Apply your naming convention uniformly across the dataset:
Do not just fix rows that look obviously wrong. Apply rules to the entire dataset, including records that appear clean. Subtle inconsistencies are the hardest to find and the most damaging.
Normalization alone does not eliminate duplicates; it just makes them more visible. Now use:
When you find duplicates, decide which record to keep, typically the most complete and most recently verified, and merge any unique data before deletion.
Do not push changes live without a human review pass. Have a subject-matter expert check 5–10% of cleaned records. Pay extra attention to:
One-time cleaning degrades fast. Protect your work by building prevention into your data entry process:
These are the product name cleaning best practices used by leading data teams across retail, healthcare, and manufacturing in 2026.
Every department, procurement, logistics, finance, and marketing, must pull product names from one authoritative master list. No local versions. No department-specific spreadsheets. One list, one standard.
Decide whether you write “Tablet” or “Tab”, “Milligram” or “mg”, “Solution” or “Sol”. Then document your choices and enforce them everywhere. Inconsistent abbreviations are the single biggest driver of duplicate product records.
Choose a style and enforce it globally. Title Case is the most readable and professional format for product catalogs.
Example — the same product, three ways:
Standardize the order of descriptors within every product name. A reliable format for most catalogs:
[Generic/Common Name] + [Strength or Size] + [Form or Type] + [Pack Size]Real-world examples:
Ibuprofen 400mg Tablet 10sMicrofiber Flat Mop Head 40cm Color-CodedSodium Hypochlorite 1000ppm Surface Disinfectant 5LConsistent structure makes names scannable, sortable, and searchable.
Unless your system explicitly requires them, remove symbols like &, @, #, %, and /. They break URLs, barcodes, and API requests. The one exception: hyphens and parentheses used in standardized chemical or product nomenclature.
For healthcare and chemical products, your product name should include identifiers that support audit trails:
Example:
Bleach SpraySodium Hypochlorite 1000ppm Surface Disinfectant – CDC Approved – Reg. No. 12345This level of specificity prevents confusion and speeds up regulatory reviews.
When you update your naming convention, version the document. “Product Naming Standard v2.1 — May 2026” is clear and traceable. Archive previous versions. This is non-negotiable in regulated industries where documentation and audit trails are reviewed.
Many teams maintain a downloadable product name cleaning best practices PDF for offline reference and onboarding. Publish yours internally and share it with any third-party vendors who submit product data.
Rules are only as good as the people following them. Run a short training session when you introduce new standards. Show real before-and-after examples. Make it easy to look up the standard without asking a colleague.
In 2026, traditional fuzzy matching is no longer enough. Modern AI tools use LLM-based embeddings to detect semantic duplicates — records that describe the same product in completely different words.
For example:
Tools like Atlan, WinPure, and Ovaledge now include this capability as standard. Use it.
The biggest mistake organizations make is treating product name cleaning as a one-time fix. Data drifts. New suppliers send messy data. Staff turn over. Build cleaning into your quarterly data governance cycle, not just your annual spring clean.
If you work in a regulated sector, your product name cleaning process must align with recognized frameworks.
The PIC/S Guide to Good Practices for the Preparation of Medicinal Products in Healthcare Establishments is the gold standard for naming and labeling in pharmacy and clinical settings. Key requirements include:
Its companion document, the PIC/S Guidelines for Sterile Manufacturing PDF, adds naming requirements specific to clean rooms and aseptic preparation areas — including how to reference active ingredient concentrations, preparation dates, and expiry information within a product name.
Both documents are available through the PIC/S secretariat and are essential references for any healthcare data team.
The CDC maintains guidance on CDC approved cleaning products for healthcare infection control. When cataloging these products, always include:
Before and after example:
Bleach SolutionSodium Hypochlorite 1000ppm Surface Disinfectant – CDC Approved – EPA Reg. 67619-32This format eliminates ambiguity during procurement and compliance audits.
For cleaning chemicals cataloged in a healthcare or industrial setting, OSHA’s HCS requires product identifiers that include:
Your product name and supporting data fields must support Safety Data Sheet (SDS) lookup and compliance.
One of the most common product naming tasks in healthcare data management is standardizing names for cleaning equipment and materials. Below is a reference list modeled on best-practice naming conventions.
| Equipment | Standard Product Name Format |
|---|---|
| Mop and bucket | Mop Set – Hospital Grade – 10L Capacity |
| Microfiber mop head | Microfiber Flat Mop Head – 40cm – Color Coded |
| Floor scrubber | Auto Floor Scrubber – 45cm Path – Electric |
| Steam cleaner | Steam Disinfection Unit – 1500W – Floor/Surface |
| HEPA vacuum cleaner | HEPA Vacuum Cleaner – 18L – Wet/Dry |
| Electrostatic sprayer | Electrostatic Disinfectant Sprayer – 2L Capacity |
| Pressure washer | Cold Water Pressure Washer – 120 Bar |
| Clinical waste trolley | Clinical Waste Trolley – 60L – Stainless Steel |
| Color-coded cleaning kit | Color-Coded Cleaning Kit – Zone-Specific – 5-Piece |
| Dispensing station | Wall-Mount Disinfectant Dispensing Station – 1L |
Many facilities maintain a hospital cleaning equipment list with pictures for staff training and procurement. These visual guides pair the standardized product name with a photo, dramatically reducing picking errors.
A hospital cleaning materials list PDF is another standard reference document. It should include:
Use the naming format in the table above as your template. Pair it with a photo catalog and you have a complete onboarding and compliance resource.
Manual cleaning at scale is slow and unreliable. These tools reflect the 2026 standard for product name cleaning.
rapidfuzz replaces the older fuzzywuzzy library and is significantly faster.A PIM system is the long-term infrastructure solution. It acts as your master product list, enforces naming rules at entry, and distributes clean data to every connected system.
Top PIM platforms for 2026:
When stress-testing a new naming convention, you need to throw unusual inputs at it. Data teams sometimes use name generation tools to surface edge cases — unusual characters, very long strings, or unexpected formats that break validation rules.
A random name generator is a surprisingly effective way to stress-test character limits, special-character handling, and format validation rules before your naming standard goes live.
Even experienced data teams make these errors. Recognizing them early saves months of rework.
If you clean data before writing down your naming rules, you create a new inconsistency layer on top of the old one. Define standards first. Always.
New entries get cleaned. The 80,000 records from 2019 stay untouched. Legacy data poisons your clean catalog every time someone queries it. Legacy data must be in scope.
Open text fields invite chaos. Use dropdowns, controlled vocabularies, and character-limit enforcement wherever possible. Prevention costs a fraction of what correction costs.
Normalizing formats without running semantic deduplication misses the point. Two records can be perfectly formatted and still describe the same product. In 2026, LLM-based tools make semantic deduplication accessible. Use them.
AI-generated transformations are powerful but not infallible. Always inspect and test AI-suggested changes before applying them to production catalogs — especially for regulated products. AI accelerates the work; human review ensures accuracy.
Data drifts. Staff turn over. New suppliers arrive. Product naming standards that are not actively enforced degrade within months. Build ongoing validation and quarterly audits into your governance cycle.
A well-maintained naming standard should be packaged as a downloadable PDF for internal reference and vendor onboarding.
A complete product name cleaning best practices PDF should include:
Published external resources worth downloading:
These documents should sit alongside your internal naming standard as reference material for anyone working with product data in regulated categories.
Cleaning fixes errors typos, rogue characters, extra spaces. Standardization applies a consistent format capitalization rules, abbreviation conventions, structural order. You need both. Always clean before you standardize.
At minimum, run a full audit quarterly. For catalogs with frequent new entries, set up real-time validation at the point of entry. High-volume, high-risk catalogs (pharma, medical devices) should run automated checks on every new submission.
A semantic duplicate is when two records describe the same product using different words or structures — for example, "Nitrile Exam Glove Medium Blue" and "Blue Medium Nitrile Examination Glove." Traditional exact and fuzzy matching miss these. In 2026, LLM-based tools catch them. This matters because semantic duplicates inflate your catalog, distort analytics, and cause procurement errors.
Yes. Healthcare teams should reference PIC/S guidelines and CDC recommendations. Retail and supply chain teams should align with GS1 and ISO 8000. Pharmaceutical manufacturers should follow their regulatory agency's requirements plus any internal standards tied to product registration. Chemical products in workplaces must meet OSHA HCS labeling requirements.
OpenRefine remains the best free option for visual, no-code cleaning. For teams with coding capability, Python with pandas and rapidfuzz is the most powerful free stack.
A strong foundation in naming principles helps across all domains — from product catalogs to brand naming. Understanding how to choose realistic, clear character names teaches the same core principles of clarity, uniqueness, and consistency that apply to product naming. The funny-names.org resource hub also covers broader naming strategy that translates well to data catalog work.
Maintain your primary name in your base language. Store translations as separate attributes, not as name variants. This keeps your primary catalog clean while supporting multilingual operations across regions.
In 2026, dirty product names are not just an operational inconvenience; they are an AI readiness failure. Organizations that feed messy, inconsistent product data into analytics platforms and machine learning pipelines get unreliable outputs. The garbage-in, garbage-out rule has never been more consequential.
The product name cleaning best practices in this guide give you a proven, current framework:
Your product data is one of your most valuable business assets. In 2026, clean product names are the foundation of accurate AI, reliable reporting, and seamless compliance.
Start the audit today. The cost of waiting only compounds.
Want to sharpen your naming instincts further? Check out the top name generators for gamers and streamers, an unexpectedly useful resource for testing how naming systems handle edge-case inputs, unusual character combinations, and format extremes before your validation rules go live.









Funny Names Generator brings laughter and creativity together create random, unique, and funny names anytime, anywhere for free.
Popular Tools
Copyright © 2026 Funny Names Generator. All Rights Reserved | Developed by Ahsan Mushtaq